11.2 Oxygen Supply
Oxygen used for welding is stored in cylinders of various sizes, which are usually painted. Since there is no standard national code, the cylinders can be painted any color selected by the manufacturer.
Oxygen is stored in cylinders at pressures from 2000 psig to 2640 psig (pounds per square inch gauge), or 13790kPa to 18202kPa (kilopascals). The pressure varies, depending on the cylinder material, size, and room temperature.
Oxygen can be obtained through various processes. One process consists of the liquefying of atmospheric air by compression and cooling (called liquefaction). Atmospheric air consists of approximately 21% oxygen, 78% nitrogen, 1% other gases (by volume). The liquefaction of air is used to obtain oxygen and nitrogen and other atmospheric noble gases. This is done by separating each of the various elements through fractional distillation in special cryogenic air separation units. This distillation is achieved through a series of compressions and expansions to reach very low temperatures.
Since oxygen and nitrogen condense at different temperatures, it is easy to separate the oxygen from the nitrogen by means of fractional distillation. Oxygen condenses at the higher temperature of –297°F (–183°C) and nitrogen condenses at –320°F (–196°C) at 1 atm. After being separated, the liquid oxygen and nitrogen can be placed in separate Dewar flasks for storage and shipment. As liquid gases are warmed, they become gaseous and can then be stored under pressure in thick-walled cylinders.
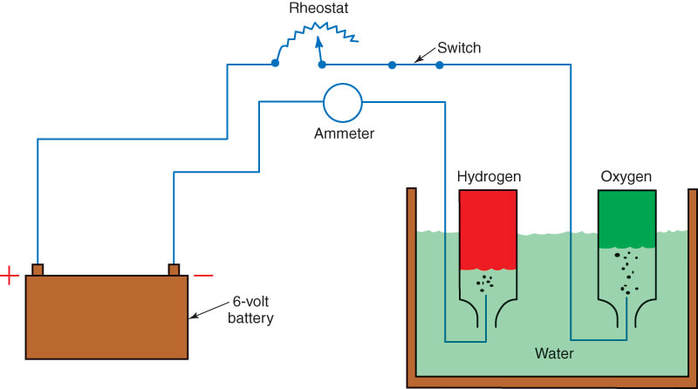
Another process used to produce oxygen is the electrolysis of water. In this process, an electric current is passed through water causing the water (H2O) to separate into its elements, which are oxygen (O) and hydrogen (H). In the electrolytic process, oxygen will collect at the positive electrode, and hydrogen will collect at the negative electrode. Figure 11-3 illustrates an experimental schematic of the electrolysis of water.
Commercial oxygen is close to 100% pure. A popular method of distribution consists of shipping oxygen as a liquid. Liquid oxygen installations are chiefly used in steel mills and by steel fabricators who use large quantities of oxygen.
Oxygen sold in liquid form is available in large thermos bottle-like tanks or vessels known as Dewar flasks. Liquid oxygen is not held under high pressure. The pressure in a liquid oxygen vessel is seldom greater than 240 psig (1655kPa). The evaporation of some of the liquid keeps the temperature of the liquid very low, approximately –297°F (–183°C). At this low temperature, the oxygen remains a liquid at any pressure above atmospheric pressure. Liquid oxygen in a container will rapidly evaporate if the pressure is reduced or if the temperature is increased.
Portable liquid oxygen vessels have grown in popularity in recent years. The advantages of using just one liquid oxygen vessel instead of many oxygen cylinders is that it can be stored in a small space, and provides a far greater volume of gaseous oxygen. Figure 11-4 shows a Dewar flask, a vessel used to store liquid oxygen.
Oxygen is stored in cylinders at pressures from 2000 psig to 2640 psig (pounds per square inch gauge), or 13790kPa to 18202kPa (kilopascals). The pressure varies, depending on the cylinder material, size, and room temperature.
Oxygen can be obtained through various processes. One process consists of the liquefying of atmospheric air by compression and cooling (called liquefaction). Atmospheric air consists of approximately 21% oxygen, 78% nitrogen, 1% other gases (by volume). The liquefaction of air is used to obtain oxygen and nitrogen and other atmospheric noble gases. This is done by separating each of the various elements through fractional distillation in special cryogenic air separation units. This distillation is achieved through a series of compressions and expansions to reach very low temperatures.
Since oxygen and nitrogen condense at different temperatures, it is easy to separate the oxygen from the nitrogen by means of fractional distillation. Oxygen condenses at the higher temperature of –297°F (–183°C) and nitrogen condenses at –320°F (–196°C) at 1 atm. After being separated, the liquid oxygen and nitrogen can be placed in separate Dewar flasks for storage and shipment. As liquid gases are warmed, they become gaseous and can then be stored under pressure in thick-walled cylinders.
Another process used to produce oxygen is the electrolysis of water. In this process, an electric current is passed through water causing the water (H2O) to separate into its elements, which are oxygen (O) and hydrogen (H). In the electrolytic process, oxygen will collect at the positive electrode, and hydrogen will collect at the negative electrode. Figure 11-3 illustrates an experimental schematic of the electrolysis of water.
Commercial oxygen is close to 100% pure. A popular method of distribution consists of shipping oxygen as a liquid. Liquid oxygen installations are chiefly used in steel mills and by steel fabricators who use large quantities of oxygen.
Oxygen sold in liquid form is available in large thermos bottle-like tanks or vessels known as Dewar flasks. Liquid oxygen is not held under high pressure. The pressure in a liquid oxygen vessel is seldom greater than 240 psig (1655kPa). The evaporation of some of the liquid keeps the temperature of the liquid very low, approximately –297°F (–183°C). At this low temperature, the oxygen remains a liquid at any pressure above atmospheric pressure. Liquid oxygen in a container will rapidly evaporate if the pressure is reduced or if the temperature is increased.
Portable liquid oxygen vessels have grown in popularity in recent years. The advantages of using just one liquid oxygen vessel instead of many oxygen cylinders is that it can be stored in a small space, and provides a far greater volume of gaseous oxygen. Figure 11-4 shows a Dewar flask, a vessel used to store liquid oxygen.